The European Commission explain the details of how a future system for medical device nomenclature will help to support the EU’s new device and IVD regulations, as well as the Eudamed database, which allows for information to be exchanged between national competent authorities and the Commission. The MDR-mandate Eudamed go-live date was set for 25 March 2020.
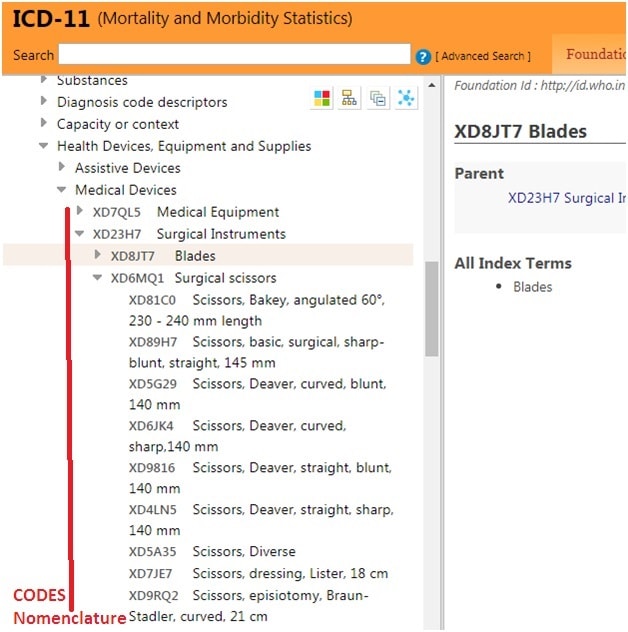
The medical device nomenclatures internationally recognized at the time of the date of application of the Regulations. In this context, global harmonization principles and orientations followed and adopted by the International Medical Device Regulators Forum (IMDRF) and the World Health Organization (WHO), are taken into particular account.
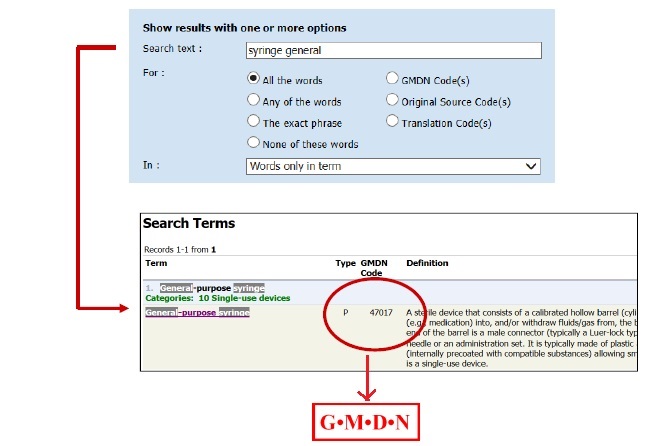
The EC said about the correspondence between the nomenclatures will be visible to operators and incorporated in the future database.This will allow all operators registering their device to find CND (National Classification of Medical Devices) nomenclature equivalent to a GMDN code (Global Medical Device Nomenclature). Next steps include establishing an MDCG subgroup for oversight of the EU nomenclature system, exploring ways to support the nomenclature work of WHO and providing additional details on the system’s governance and operation.
The nomenclature aims to not only support the functioning of Eudamed, but also to standardize descriptions in and interpretations of its information to identify and monitor incidents and root causes.
The commission said about the structure and design of the future nomenclature should facilitate the establishment of links with the codes defining Notified Bodies competence (designation scope), the scope of medical devices QMS (Quality Management System)/QA (Quality Assurance) certificates, and product portfolios in the mandate of Authorized Representatives.
Nomenclature shall be available free of charge to manufacturers and other natural or legal persons required by the Regulation to use that nomenclature, this meaning that no manufacturer or natural/legal person should be subject to fee or suffer from any discrimination, compared to other operators, in relation to the use of the nomenclature under the new Medical Device Regulations. It shall be therefore ensured that relevant names and codes are accessible (in full) in Eudamed to all operators that are requested to provide the relevant UDI submissions. UDI is a key for device nomenclature while registering and or updating device in real time scenarios.
Eudamed also “shall make available the most updated names/codes related information, to the benefit of operators and general public,” Therefore, the nomenclature provider will need to have procedures and services in place that shall allow Eudamed to be kept up-to-date at any time. When setting the procedure, in particular the frequency, related to the periodic review of nomenclature terms and descriptions, this shall be taken into account.”
Manufacturers/Economic operators (EO) shall keep ready with all the details on Device registration, UDI registration, certificates and notified bodies, clinical investigational performance, Vigilance and Market surveillance and support with Eudamed requirements.
What to do for Device Nomenclature codes in Eudamed:
- Ensure to register and update codes with Eudamed in UDI part
- Ensure to check codes as per GMDN, WHO/CND codes?
- Ensure to check device nomenclature codes for EC approved devices
- Keep up to date & Coordinate with NBs and Eudamed
- Periodic review for medical device code, investigational studies, vigilance and market surveillance.
Sample Nomenclature:
1.WHO ICD-11 codes on Medical device (EU adopted)
2. IMDRF: GMDN codes on Medical device. (EU adopted)
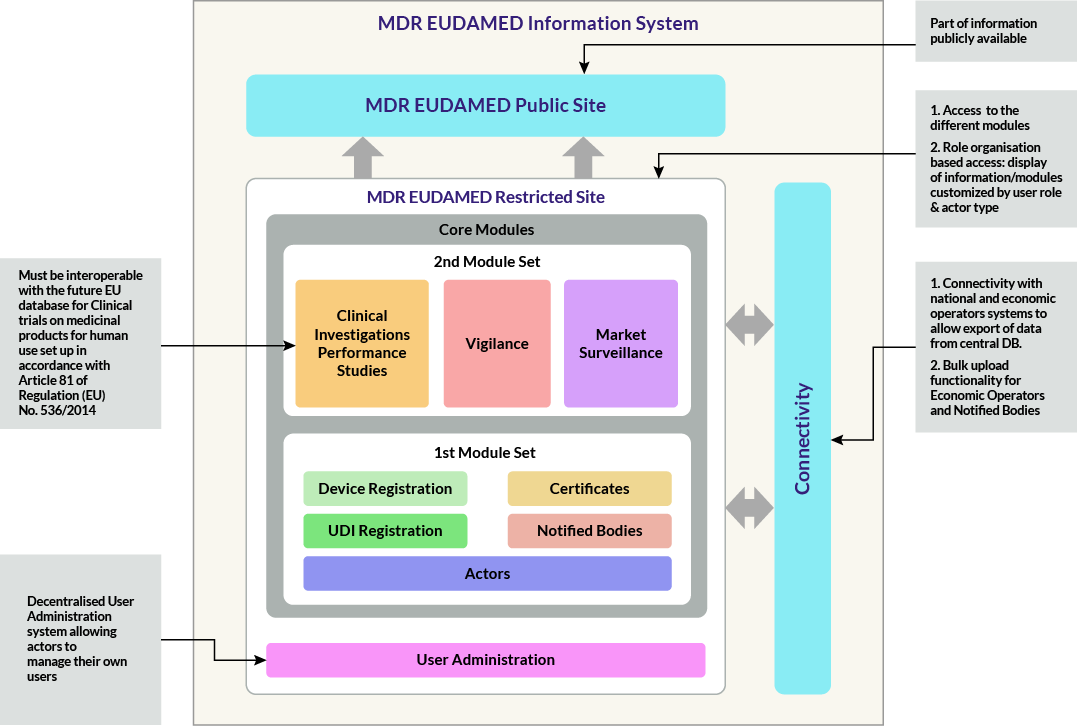
EUDAMED System functions & Connections:
Eudamed will be focusing information from Economic Operators/manufacturers on the device registration, UDI registration, certificates and notified bodies’ information. But it is important to remember that Eudamed is intended to span the entire regulatory lifecycle. UDI information is inherited throughout the process, and shall be connected to Certificates (connections made by NBs), Clinical Investigations, Vigilance Reports and Post Market Surveillance. Manufacturers are not able to responsible for entering or maintaining data at all these levels in single step. But linking tools are very important in effectively connecting information across the system.
Manufacturers/Economic operators shall be register in the database, Eudamed designed by different modules as designed by organization based and customized by user role and actor type. The functional specifications are divided into two types MDR Eudamed restricted website and MDR Eudamed public websites.
Restricted website contains two core modules,
- 1st Module set: Manufacturers or EO are needed to submit/register the information for Device registration, UDI registration, certificates and notified bodies.
- 2nd module set: Manufacturers or EO are needed to submit/register the information for clinical investigational performance, Vigilance and Market surveillance.
Roles of interlinking tools:
Nowadays, the connecting interlinking tools will be act as important roles to minimize efforts and providing greater access and operate with a larger scope.These tools are not only support with manufacturer’s specific requirements but also support with MDR standards.
The interlinking tools should be capture the below information for Eudamed.
- Understand the Eudamed requirements
- Support the data as per MDR/IVDR regulations
- Maintain Data for 1st Module set & 2nd module sets
- Keep up to date update during periodic review of device
- Data management
- Version control



