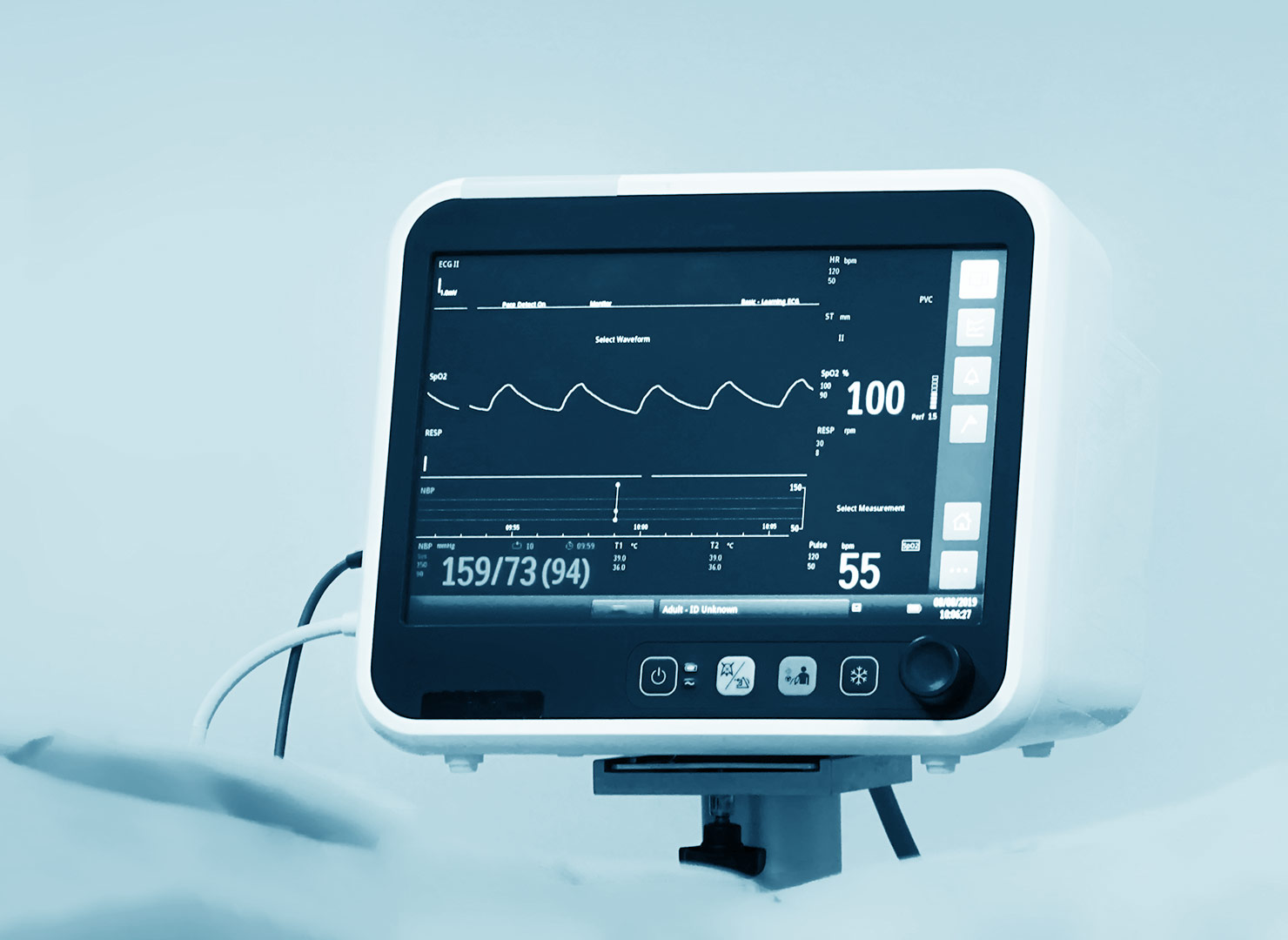

Webinar: EU IVDR Performance Evaluation, Data Requirements & Gaps
As some of you are aware, In Vitro Diagnostic Regulation (IVDR) will be effective May 2022. As this regulation has several new requirements, some simple and some complex.

Webinar: EU IVDR Performance Evaluation, Data Requirements & Gaps
As some of you are aware, In Vitro Diagnostic Regulation (IVDR) will be effective May 2022. As this regulation has several new requirements, some simple and some complex.

Webinar: Medical Device PMS & PMCF: Challenges & Opportunities
The new 2017/745/EU Medical Device Regulations (MDR-2020) require increased emphasis on Post- Market Clinical Follow-Up (PMCF


Webinar: Importance & Requirements of Clinical Evaluation for CER
The revised guidance MEDDEV 2.7/1 rev 4 reveals a course of changes to a more prescriptive and demanding set of expectations.


Webinar: Growing need of Clinical Data, PMCF & Registries for Devices
Post Market Clinical Follow Up (PMCF) studies are a critical component of the clinical evidence required to comply with the new European regulations.

Webinar: Device Compliance with MEDDEV 2.7.1 Rev 4 for Clin Eval / CER
Clinical evaluation is a methodologically sound ongoing procedure to collect, appraise and analyze clinical data pertaining to a medical device